 His experimental results were further investigated by Rutherford and Bohr, which further provided important insights into the atomic world.īefore directly jumping Thomson's findings, let us understand some basic knowledge on cathode rays and the cathode-ray tube. However, Thomson's contributions remain more significant than the rest. Thomson was not the only one working on cathode rays, but several other players like Julius Plücker, Johann Wilhelm Hittorf, William Crookes, Philipp Lenard had contributed or were busy studying it. The apparatus of his experiment is called the cathode-ray tube (CRT). These particles later were named electrons. In 1897, he showed that cathode rays were composed of very small negatively charged particles. 
He was well-known for the discovery of the electron. The device is called an oscilloscope which is frequently used in medical treatment.Sir Joseph John Thomson was a British physicist and Nobel Laureate. Used to measure changes in electrical voltage with time. They helped the transition from the industrial age to the digital.ģ. 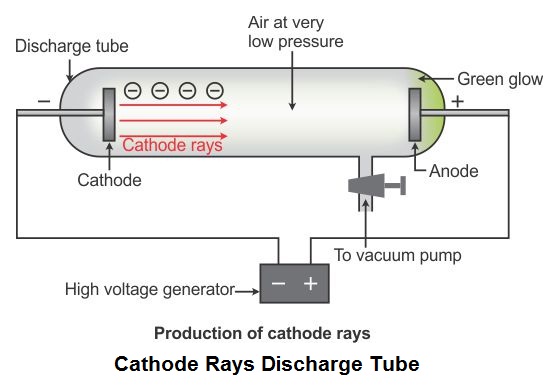
Vacuum tubes were initially used in the place of silicon transistors in electronics. In old displays, vacuum tubes were used by directing the beam of electrons using deflection plates, then the beam causes fluorescence on the screen which we see as white.Ģ. So, the cathode ray experiment is also commonly known as J.J. The above modified experiments were performed by J.J. This also helped scientists in finding the charge of electrons. Hence, energy of electron at point A = energy of electron at point B Applying a Magnetic FieldĬathode rays also get deflected from their path if a magnetic field is applied. At point B the electron stops due to the activation of stopping potential, so we apply the law of conservation of energy between the two points. When electrons move from one point to another, say from A to B. When we apply electric field in parallel but in the opposite direction to the cathode rays and if it is sufficiently high for the cathode rays to stop, then the magnitude of the applied voltage is called stopping potential. The magnitude of deviation is proportional to the magnitude of the electric field. When cathode rays hit from the cathode travel towards the anode and hit the anode, a fluorescence or glow is produced.Ĭathode ray deviates from its path due to the application of an electric field. When a high voltage is applied between the two electrodes of an evacuated discharge tube and the back of anode of the discharge tube is coated with a material, like zinc sulfide. They were later named electronsafter particles postulated by George Johnstone Stoney. Thomson measured the weight of cathode rays and showed that they were actually a beam of particles. German scientists Eilhard Wiedemann, Heinrich Hertz and Goldstein said they were some new form of electromagnetic radiation. 
Scientists Crooks and Arthur Schuster said they were electrically charged atoms. Scientists came up with two theories regarding cathode rays when they were originally discovered. Eugene Goldstein was the one who actually gave cathode rays their name. Cathode rays were first identified by a German physicist named Johann Hittorf when he realized that something was travelling through the tube. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
